Plug Flow Reactor: Principles, Features, and Applications
This guide dives into Plug Flow Reactors (PFRs), a key tool in chemical engineering that helps control chemical reactions efficiently in a tube or pipe. Unlike other reactors that mix everything up, PFRs keep everything flowing smoothly in one direction, which is great for making sure reactions happen just right. This means they can be really good at making products consistently and safely, especially in industries like pharmaceuticals, oil refining, and plastics. We'll look at how PFRs work, why they're often better than other types of reactors, and the kind of tasks they're really good at.Catalog

What is a Plug Flow Reactor?
A Plug Flow Reactor (PFR), also known as a piston flow reactor, is an important tool in chemical engineering used to study how chemical reactions happen inside a long tube. In this type of reactor, fluids are pumped in at one end and move steadily through the tube to the other end at a constant speed. The flow is smooth and even, with no swirling or mixing along the way. This means that the fluids travel in separate layers or plugs, keeping the different stages of the reaction apart. In other words, the fluids that enter the tube earlier are always ahead of the fluids that enter later, and they don’t mix with each other as they move along. This helps to control the chemical reaction because the reactants are kept in order, allowing each part of the reaction to happen step by step as the fluids flow through the tube.
The unique way that fluids move in a PFR has some important benefits for chemical reactions. Since there is no mixing backward, each layer of fluid spends the same amount of time in the reactor, leading to a consistent reaction process. This makes it easier to predict how much product will be made because the reaction time is the same for every part of the fluid. You can adjust the length and width of the tube, the speed of the flow, and the temperature to control the reaction and get the best results. Plug Flow Reactors are especially useful for reactions that need high temperatures or involve gases because the steady flow keeps the reaction under control. They are commonly used in industries like oil refining, making plastics, and treating waste gases. This design is also good for large-scale production because it’s easier to predict and control what happens inside the reactor.
Features of Plug Flow Reactors
Directional Flow
Plug Flow Reactors (PFRs) are characterized by their unidirectional flow, where reactants and products move exclusively in a forward direction along the reactor’s length. This streamlined flow pattern is achieved by designing the reactor as a tubular conduit, which minimizes any back-mixing of fluid elements. In a PFR, the fluid elements, often referred to as plugs, maintain their identity as they progress through the reactor, meaning there is minimal diffusion between adjacent plugs. This orderly movement is important for achieving precise control over reaction kinetics because each plug experiences a unique and predictable set of reaction conditions as it moves through the reactor. Reactants at the inlet encounter the highest concentrations and the freshest chemical environment, which can be strategically leveraged to drive rapid reaction rates.
Figure 2. Plug Flow Reactor (PFR) Directional Flow Representation
As the plugs advance, reactant concentrations decrease while product concentrations increase, following a sequential conversion process along the reactor’s length. This systematic progression is advantageous for reactions requiring strict stoichiometric control or those sensitive to fluctuations in concentration. By preventing recirculation and maintaining a consistent directional flow, PFRs reduce the risk of side reactions that might occur if reactants and products were to mix or recycle. This makes PFRs ideal for processes like polymerization, combustion, or complex chemical syntheses. The predictable flow pattern enhances the accuracy of reaction modeling and scale-up, making PFRs highly valuable in both research and industrial applications.
Concentration Gradient
A defining characteristic of Plug Flow Reactors is the development of a concentration gradient along the length of the reactor. At the inlet, reactants are introduced at their highest concentrations, creating a driving force for the reaction to proceed. As the reaction progresses, the reactant concentrations steadily decrease while product concentrations simultaneously increase. This results in a well-defined concentration gradient that evolves predictably from the inlet to the outlet of the reactor. This gradient is steepest near the inlet, where reactant concentrations are at their peak and the reaction rate is highest due to the abundance of reactants. As the reactants are consumed, the gradient gradually flattens, reflecting the reduced reaction rate as the system approaches equilibrium.
The absence of back-mixing in PFRs ensures that each plug experiences a unique concentration environment, promoting efficient and sequential reactant conversion. The concentration gradient is advantageous for reactions with concentration-dependent kinetics, where the reaction rate is a function of reactant concentration. In such cases, the high reactant concentration at the inlet maximizes the initial reaction rate, leading to efficient utilization of reactants. This progressive consumption of reactants minimizes the risk of undesirable side reactions that could occur at intermediate concentrations. For processes involving competitive reactions, the concentration gradient can be managed to favor the desired product pathway. This makes PFRs highly effective for complex synthesis processes, such as in the pharmaceutical or fine chemicals industry, where product selectivity and yield optimization.
Consistent Residence Time
One of the most advantages of Plug Flow Reactors is the uniform residence time distribution. In a PFR, each plug of reactant spends the same amount of time traveling through the reactor, from the inlet to the outlet. This uniform residence time ensures that all reactant molecules are subjected to identical reaction conditions, including temperature, pressure, and concentration profiles. This consistency is achieved because the plugs move through the reactor in an orderly, sequential fashion without any back-mixing or radial dispersion. Consequently, there is no variation in the time reactants spend inside the reactor, which is in stark contrast to Continuous Stirred-Tank Reactors (CSTRs), where reactants can have widely varying residence times due to thorough mixing.
Uniform residence time is good for maintaining consistent product quality, especially in complex chemical processes. For example, in polymerization reactions, consistent residence time ensures uniform polymer chain lengths and molecular weight distribution, leading to superior product performance. In pharmaceutical synthesis, it guarantees consistent chemical purity and potency for regulatory compliance and product efficacy. The predictable residence time distribution simplifies kinetic modeling and scale-up, enabling accurate simulation of reaction behavior on an industrial scale. This predictability enhances process control and optimization, leading to increased productivity and cost efficiency.
High Conversion Efficiency
Plug Flow Reactors are known for their high conversion efficiency per unit volume, which is superior to that of Continuous Stirred-Tank Reactors (CSTRs). In PFRs, reactants are continuously consumed as they move through the reactor, following a linear and progressive reaction pathway. This sequential consumption minimizes the concentration of unreacted species at any point along the reactor’s length, resulting in higher overall conversion rates. The absence of back-mixing allows reactants to be exposed to optimal reaction conditions throughout their journey, maximizing the reaction rate and minimizing the formation of unwanted by-products. This is advantageous for fast or highly exothermic reactions that require rapid reactant depletion to maintain safety and product integrity.
In such cases, the plug flow design facilitates effective heat management by preventing localized temperature spikes that could lead to thermal runaway or product degradation. The high conversion efficiency of PFRs reduces the required reactor volume, leading to smaller equipment footprints and lower capital costs. This efficiency also translates into reduced operational expenses, as less energy and fewer raw materials are needed to achieve the desired product yield. This makes PFRs attractive for large-scale industrial applications, such as petrochemical processing, bulk chemical production, and environmental treatment systems.
Steady-State Operation
Plug Flow Reactors operate under steady-state conditions, maintaining consistent flow rates, concentration profiles, and temperature distributions throughout the reactor. In steady-state operation, the inflow and outflow rates are constant, ensuring a continuous and uninterrupted production process. This stability is good for maintaining uniform product quality, which is important in industries such as petrochemical refining, polymer manufacturing, and large-scale chemical synthesis. The steady-state nature of PFRs simplifies process control and automation because the system’s operating parameters remain constant over time.
This predictability enhances scalability and process reliability, reducing the complexity of monitoring and adjusting reaction conditions. Moreover, steady-state operation minimizes fluctuations in product composition, ensuring consistent chemical purity and quality for high-value products like pharmaceuticals and specialty chemicals. Steady-state operation also facilitates energy efficiency and process optimization, as the constant operating conditions allow for precise control of thermal and mass transfer processes. This makes PFRs suitable for applications requiring tight control over reaction kinetics and product selectivity, further contributing to cost savings and sustainability in chemical manufacturing.
Efficient Heat Transfer
Plug Flow Reactors are designed to support efficient heat transfer and managing temperatures in exothermic or endothermic reactions. The consistent flow pattern prevents back-mixing, allowing for precise temperature control along the reactor’s length. This enables the implementation of controlled temperature gradients or the use of heat exchangers to optimize thermal management. Effective heat transfer not only improves energy efficiency but also enhances product selectivity, making PFRs ideal for temperature-sensitive reactions, such as those in fine chemical synthesis or catalytic processes. In highly exothermic reactions, PFRs help dissipate heat effectively, preventing thermal runaway and ensuring safe operation. Conversely, in endothermic reactions, they facilitate efficient heat supply to maintain reaction rates and conversion efficiency.
Versatility Across Phases
PFRs are versatile and can accommodate gas-phase, liquid-phase, and heterogeneous reactions, including those involving catalysts. This adaptability allows them to be used in a wide variety of chemical processes, such as gas-phase oxidation, liquid-phase polymerization, and catalytic hydrogenation. The plug flow design maximizes reactant-catalyst contact, enhancing reaction rates and catalyst efficiency for industrial applications in petrochemical processing, polymer manufacturing, and environmental catalysis.
Schematic of Plug Flow Reactor
A Plug Flow Reactor (PFR) is designed to be efficient and useful in chemical processing. It usually looks like a long, narrow tube or pipe, which lets chemicals flow steadily from one end to the other. Inside the tube, there are no mixing tools, like stirrers. Because of this, the chemicals move smoothly, like a piston, without mixing together. This steady flow helps the reactions happen evenly and allows for good control over how fast they occur. This makes PFRs very useful in chemical factories and industries.
Figure 3. Plug Flow Reactor Schematic
The diagram depicts a Plug Flow Reactor (PFR), a type of chemical reactor designed for continuous chemical processing. In this setup, reactants enter at the inlet (x = 0) and move steadily through a cylindrical tube toward the outlet (x = L). The flow within the reactor is streamlined, maintaining a consistent velocity profile where each fluid element travels parallel to the reactor’s axis without any back-mixing. This arrangement allows reactants to gradually convert into products as they progress along the reactor’s length. The shaded segment marked as 𝑑𝑥 represents a tiny volume element within the reactor. This differential slice is needed for modeling chemical reactions occurring inside the PFR, as it helps evaluate changes in reaction rate and concentration as the reactants advance.
The absence of radial mixing preserves the plug flow condition, ensuring that each fluid element maintains its identity and composition throughout its journey. As a result, concentration and reaction rates vary along the reactor's length but remain uniform across any cross-section. This plug flow characteristic makes PFRs suitable for chemical processes that require precise control over reaction time and product formation. Since all fluid elements have the same residence time, the reactor delivers consistent product quality and efficient reactant conversion. Due to these advantages, PFRs are widely used in chemical manufacturing, including applications such as polymerization, gas-phase reactions, and catalytic processes.
Operating Principle of Plug Flow Reactor
A Plug Flow Reactor (PFR) operates on a unique flow pattern that keeps reaction conditions consistent throughout its length. This design allows precise control over residence time, temperature, and pressure, making PFRs highly efficient for chemical processes requiring accurate reaction management. This section explores the detailed operational principles of PFRs and their importance in industrial chemical processing.
Continuous and Uniform Movement
In a Plug Flow Reactor (PFR), fluids move continuously and uniformly along a cylindrical tube or pipe, creating a highly organized flow pattern. Reactants are introduced at one end of the reactor and flow steadily through to the other end, where they exit as products. This design ensures a seamless progression from inlet to outlet without any interruption in the movement of the fluid. The reactor’s architecture maintains a consistent flow direction, strictly preventing any backward mixing or recirculation. This unidirectional movement is important for preserving the chemical integrity of the reactants and products as they travel through the reactor. A key characteristic of this flow pattern is that each small segment, or plug, of fluid retains its own unique chemical identity as it moves through the reactor. These plugs do not mix with each other laterally, ensuring that the composition within each plug remains constant throughout its journey.
This isolation of fluid segments preserves the concentration gradients for efficient chemical reactions. The absence of radial mixing also leads to a more predictable reaction profile, allowing for accurate modeling and optimization of chemical processes. The streamlined flow pattern within a PFR minimizes turbulence and chaotic movement, which helps maintain the integrity of reactant streams. This orderly flow supports high conversion efficiency because the reactants consistently experience the same reaction conditions as they pass through the reactor. The absence of back-mixing ensures that the residence time of each plug is identical, leading to uniform exposure to temperature and pressure. This uniformity enhances reaction rates and improves product selectivity, resulting in a more controlled and efficient chemical process. Due to the precise control over flow patterns and reaction conditions, PFRs are suitable for reactions that require a consistent environment to achieve high yields and selectivity.

Figure 4. Plug Flow Reactor (PFR) Continuous Flow and Reaction Progression
The reactor’s ability to maintain steady-state operation with minimal fluctuations in concentration or temperature makes it an ideal choice for chemical and pharmaceutical industries. In these sectors, maintaining product consistency and quality is important, and the predictable performance of PFRs supports these requirements effectively.The scalability of PFRs further enhances their utility in industrial applications. Their modular design allows for easy adjustment of reactor length and diameter to accommodate different production capacities. This flexibility, combined with their efficient flow characteristics, makes PFRs a preferred option for large-scale continuous processing. As a result, they are widely used in processes such as polymerization, hydrogenation, and catalytic reactions, where maintaining a controlled reaction environment is need for product quality and process efficiency.
Advantages in Chemical Transformations
Plug Flow Reactors (PFRs) offer several advantages for chemical transformations. They provide precise reaction control by maintaining a steady flow rate and preventing back-mixing, ensuring that each plug of reactant experiences consistent reaction conditions. This enhances efficiency and minimizes by-products, which is important for sensitive reactions like alcohol oxidation or pigment production. PFRs also deliver high product purity and consistency since all reactants undergo the same reaction time and conditions. By preventing back-mixing, they improve selectivity and reduce unwanted side reactions, making them ideal for complex chemical processes. Additionally, PFRs support continuous production, minimizing downtime and reducing costs, which enhances efficiency in large-scale manufacturing. Their scalability and flexibility allow for easy adjustment of flow rates and reactor dimensions, ensuring consistent results from pilot-scale to full-scale production. These advantages make PFRs a preferred choice for controlled chemical transformations in industries requiring high precision and efficiency.
Uniform Flow and Reaction Progression
Plug Flow Reactors (PFRs) are designed to maintain a uniform flow, ensuring that all reactant particles travel through the reactor at the same velocity. This uniformity guarantees that each infinitesimal plug of fluid, containing reactant molecules, identical residence times as it moves from the inlet to the outlet. As a result, every molecule entering the reactor is exposed to consistent reaction conditions, including temperature, pressure, and concentration gradients, as it progresses through the reaction pathway. This uniform flow pattern minimizes variations in reaction rates, leading to more predictable and controllable chemical transformations. And because there is no lateral mixing within each plug, the concentration profile remains sharply defined along the reactor’s length, allowing for precise control over reaction kinetics and conversion rates. This characteristic makes PFRs advantageous for conducting reactions that require strict residence time management to achieve optimal yields.
The plug flow pattern in PFRs effectively eliminates back-mixing or recirculation of reactants, a common issue in other reactor types such as Continuous Stirred Tank Reactors (CSTRs). By preventing any interaction between reactants at different stages of conversion, PFRs enhance reaction efficiency and ensure a more uniform product composition. This lack of back-mixing also minimizes the formation of undesired by-products, as reactants cannot prematurely interact with more advanced reaction intermediates or final products. The resulting uniform reaction environment is beneficial for complex chemical systems for selectivity and product purity. Consequently, PFRs are widely used in industries requiring high-precision chemical synthesis, such as pharmaceuticals and specialty chemicals. Their ability to maintain consistent reaction conditions while avoiding cross-contamination between reaction stages shows their importance in modern chemical engineering processes.
Optimization of Reaction Kinetics
In a Plug Flow Reactor (PFR), the time that each group of reactant molecules spends inside the reactor is consistent, which is very important for getting the best reaction rates and high conversion efficiency. In a PFR, reactants enter as separate plugs that move through the reactor at the same speed, meaning each plug experiences the same conditions, such as temperature and pressure, as it travels through. This even exposure allows for precise control over how far the reaction goes and what products are made. Because every plug gets the same treatment, the chances of unwanted side reactions or variations in product quality are greatly reduced. This makes PFRs useful for reactions that are sensitive to changes in temperature, pressure, or time. By providing a steady and predictable environment, PFRs help achieve consistent and reliable reaction results, leading to optimized reaction rates.
PFRs also prevent mixing of reactants along the flow path, which helps in controlling the reaction more effectively. Unlike Continuous Stirred Tank Reactors (CSTRs), where the reactants are continuously mixed, PFRs maintain a steady flow where reactants move forward without mixing with the plugs behind or ahead of them. This ensures that the reactants are gradually converted into products as they move through the reactor, which helps avoid overreaction or underreaction. This precise control over reaction time and concentration improves the selectivity for the desired product, resulting in higher yields and better overall efficiency. This level of control is important for complex reactions that produce intermediate species, as it allows careful management of these intermediates to avoid unwanted side reactions. Because of this, PFRs are widely used in industries where high selectivity, productivity, and consistent product quality are required.
Uniform Temperature and Pressure Profiles
The steady flow pattern characteristic of Plug Flow Reactors (PFRs) ensures uniform temperature and pressure profiles throughout the entire length of the reactor. This uniformity is good in preventing localized hot spots or cold zones that could negatively impact reaction rates and product quality. In chemical processes, temperature variations can lead to uneven reaction rates, causing the formation of undesirable by-products or inconsistent product yields. By maintaining a consistent temperature profile, PFRs enable more precise control over reaction kinetics, ensuring that the chemical reactions proceed at the intended rate. This control is beneficial for highly exothermic or endothermic reactions, where even minor temperature fluctuations can lead to runaway reactions or incomplete conversions. The uniform temperature distribution contributes to enhanced safety, product consistency, and overall process reliability.
In addition to temperature uniformity, the steady flow pattern in PFRs also ensures consistent pressure profiles along the reactor’s length. A stable pressure environment helps maintain the desired flow rates and reactant concentrations for achieving optimal conversion efficiency. This consistency enhances heat and mass transfer efficiency, allowing reactants to interact evenly with the catalyst or reaction medium. The improved mass transfer minimizes concentration gradients, which can otherwise limit reaction rates and lower conversion efficiency. Uniform pressure and temperature profiles reduce mechanical stress on the reactor walls, prolonging equipment life and reducing maintenance costs. Altogether, the ability of PFRs to maintain steady temperature and pressure conditions maximizes reaction efficiency and product quality while ensuring safe and reliable operation.
Control Over Reaction Selectivity
Plug Flow Reactors (PFRs) provide advantages in controlling reaction selectivity due to their consistent flow pattern and the absence of back-mixing. In a PFR, reactants move through the reactor as a plug, maintaining their relative positions throughout the length of the reactor. This unidirectional flow pattern allows for a predictable progression of the chemical reaction, as reactants are steadily consumed along the reactor’s length. As the reactants are depleted, a concentration gradient is naturally established, which continuously drives the reaction forward. This gradient ensures that the reactants are exposed to optimal reaction conditions at each point along the reactor, promoting efficient conversion rates. The orderly flow minimizes the likelihood of premature mixing of reactants and products, preserving the intended reaction pathway and enhancing selectivity. The absence of back-mixing not only contributes to better control over the reaction environment but also helps maintain a sharper transition between reactant-rich and product-rich zones, optimizing the overall reaction efficiency.
This flow characteristic is advantageous for reactions that follow first-order or higher-order kinetics, where the reaction rate is directly influenced by reactant concentrations. In such cases, the concentration gradient along the reactor length ensures that the reaction rate is consistently high where reactant concentrations are greatest, tapering off as reactants are depleted. The lack of radial mixing in PFRs guarantees that all reactant molecules are uniformly exposed to the catalyst or reaction conditions, preventing localized variations that could lead to side reactions or the formation of undesired by-products. This uniform exposure enhances the selectivity towards the desired product, as it reduces the chances of competing reactions that occur under varying concentration conditions. By maintaining a controlled environment and preventing deviations in reactant concentrations, PFRs offer superior control over reaction pathways, maximizing yield and product purity.
Comparison with Continuous Stirred Tank Reactor (CSTR)
Choosing between a Continuous Stirred Tank Reactor (CSTR) and a Plug Flow Reactor (PFR) depends on the specific needs of a chemical process. These two reactor types have distinct operational principles and are suited for different reaction kinetics. Understanding their differences helps in selecting the most efficient reactor for a given application.
Ideal Mixing vs. Segmented Flow Dynamics
CSTRs (Continuous Stirred Tank Reactors) are designed to achieve and maintain ideal mixing conditions, ensuring that the concentration, temperature, and pressure remain uniform throughout the entire reactor volume. This means that any reactants entering the system are instantaneously and completely mixed with the contents already inside, leading to a homogeneous mixture at all times. Because of this well-mixed nature, the output stream leaving the reactor has an identical composition to the internal reaction mixture, ensuring steady-state operation. This characteristic is beneficial for reactions that require stable operating conditions, as it allows for precise control over reaction parameters. The well-mixed environment simplifies kinetic modeling since reaction rates remain consistent across the reactor. This predictability is advantageous for exothermic reactions that pose a risk of thermal runaway, as efficient heat dissipation throughout the reactor minimizes the likelihood of localized hot spots. As a result, CSTRs are commonly used in large-scale industrial applications where maintaining a controlled and uniform reaction environment is need for both safety and efficiency.

Figure 5. Comparison of Ideal Mixing vs. Segmented Flow in Reactors
In contrast, PFRs (Plug Flow Reactors) operate under a segmented or plug flow regime, where individual fluid elements or plugs move sequentially through the reactor without mixing laterally with one another. This flow pattern establishes a concentration gradient along the reactor’s length, meaning that reactant concentrations are highest at the inlet and progressively decrease as they are consumed in the reaction, while product concentrations increase toward the outlet. Because each plug retains its distinct chemical composition throughout its journey, the reaction progresses in a controlled and predictable manner along the reactor’s axial direction. Unlike CSTRs, which assume instantaneous mixing, PFRs allow for a gradual transformation of reactants into products, mimicking the behavior of batch reactors but on a continuous basis. This flow behavior is useful in reactions where minimizing back-mixing is necessary to achieve high conversion efficiency. The absence of complete mixing ensures that reactants spend a defined amount of time in the reactor, allowing to design reactors with optimized residence times for specific reactions.
The segmented flow characteristics of PFRs offer advantages for reactions requiring precise control over reaction kinetics and product selectivity. By preventing the dilution of intermediate species through mixing, PFRs enable better manipulation of reaction pathways for optimizing selectivity and minimizing the formation of unwanted by-products. This is important in industries such as petrochemicals and pharmaceuticals, where the purity of the final product is a key concern. The ability to maintain distinct chemical compositions within each plug allows for more efficient use of catalysts, as reactants encounter optimal conditions throughout their progression. While CSTRs provide the advantage of steady-state operation and ease of control, PFRs are better suited for scenarios where maximizing conversion and maintaining strict kinetic control are priorities. The choice between a CSTR and a PFR depends on the specific requirements of the reaction, including factors such as desired product yield, heat management, and reaction time constraints.
Conceptual and Operational Differences
The flow dynamics in a Plug Flow Reactor (PFR) can be conceptually understood as a series of extremely thin, perfectly mixed Continuous Stirred Tank Reactors (CSTRs) arranged sequentially along the reactor’s length. Each of these miniature CSTRs ensures complete mixing in the radial direction while maintaining a strict separation in the axial direction, meaning there is no backward mixing between different segments. This unique characteristic allows for the establishment of a continuous and gradual concentration gradient, where reactant concentrations steadily decrease while product concentrations increase as the reaction progresses from the inlet to the outlet. Because of this design, PFRs effectively drive chemical reactions forward with greater efficiency, ensuring that reactants undergo a controlled transformation into products without the risk of back-mixing diluting reaction progress. This segmented flow model is highly beneficial for accurately simulating complex reaction kinetics, as it allows predict reaction rates and optimize reactor dimensions based on the time-dependent evolution of reactants and products. The ability to precisely control reaction conditions along the reactor length makes PFRs useful for processes requiring specific reaction sequences, such as those involving multiple reaction steps or intermediates that need precise exposure to reactants before moving to the next phase.
On the other hand, Continuous Stirred Tank Reactors (CSTRs) function under the principle of complete and uniform mixing throughout the reactor’s entire volume, ensuring that all reactant molecules are evenly distributed and exposed to identical reaction conditions. However, this complete mixing results in a broad residence time distribution, meaning that not all reactant molecules spend the same amount of time within the reactor. Some molecules may exit the system too soon, before undergoing a complete reaction, while others may remain in the reactor for an extended period, potentially leading to overreaction or unwanted side reactions. This variability in residence time can directly impact the consistency and quality of the final product, making CSTRs less ideal for reactions that require precise residence time control or high conversion efficiency. In industrial applications where reaction uniformity and steady-state operation are needed, CSTRs remain a popular choice due to their ease of operation and ability to handle large-scale continuous production. However, for highly selective reactions or those involving multiple reaction steps, PFRs often provide superior performance by ensuring that reactants follow a well-defined and predictable reaction path without the unpredictability associated with broad residence time distributions in CSTRs.
Kinetic Control and Efficiency
Continuous Stirred Tank Reactors (CSTRs) provide a well-mixed and stable reaction environment, making them advantageous for chemical processes that involve slow reaction kinetics or require precise thermal regulation. Since reactant concentrations remain constant throughout the reactor due to continuous stirring, reaction rates can be maintained at steady levels, ensuring predictable performance. This is useful for reactions where maintaining a uniform concentration profile is necessary to sustain reaction progress, as well as for equilibrium-limited reactions where constant reactant replenishment is beneficial. The continuous agitation in a CSTR facilitates efficient heat distribution, preventing localized temperature variations that could otherwise lead to reaction instability or unwanted side reactions. By maintaining uniform thermal conditions, CSTRs minimize the risks associated with exothermic reactions, such as sudden temperature spikes that could compromise safety or product quality. This ability to regulate temperature and concentration with high precision makes CSTRs a preferred choice for large-scale industrial processes for ensuring product uniformity, minimizing side reactions, and maintaining operational safety.
In contrast, Plug Flow Reactors (PFRs) offer a distinct advantage when it comes to kinetic control and reaction efficiency for processes requiring high selectivity and predictable conversion rates. The unidirectional flow of reactants through a PFR ensures that each molecule follows a well-defined path through the reactor, experiencing a consistent residence time without back-mixing or premature dilution. This controlled progression of reactants allows for precise manipulation of reaction conditions, which is important for fast, irreversible reactions where reactants must undergo complete transformation before exiting the reactor. The absence of back-mixing also enables better control over intermediate species, ensuring that each stage of the reaction occurs under optimal conditions to maximize selectivity. By maintaining a steady concentration gradient and enabling efficient reaction progression, PFRs provide superior performance for processes that demand high conversion efficiency, making them the preferred choice for applications where maximizing yield and product purity is a top priority.
Industrial Applications and Suitability
Continuous Stirred Tank Reactors (CSTRs) are widely employed in industrial processes for continuous mixing and maintaining uniform reaction conditions. One of the most common applications of CSTRs is in fermentation processes, where microorganisms or enzymes require a consistent environment to sustain optimal metabolic activity. The constant agitation ensures that nutrients, oxygen, and other reactants are evenly distributed throughout the system, preventing localized depletion that could hinder microbial growth or enzymatic efficiency. CSTRs are highly effective for handling highly viscous fluids, such as those encountered in polymerization reactions, where maintaining uniform agitation is necessary to achieve consistent molecular weight distribution and prevent localized gel formation. Their ability to provide precise temperature control is another key factor in their industrial utility, for exothermic reactions that require efficient heat dissipation to prevent thermal runaway. The homogeneous conditions within a CSTR make it an ideal choice for liquid-phase reactions, when product quality and process stability are importance. Industries that rely on CSTRs include pharmaceuticals, fine chemicals, wastewater treatment, and food and beverage production, where consistency in reaction conditions directly impacts product performance and regulatory compliance.
Plug Flow Reactors (PFRs), on the other hand, are the preferred choice for processes that require high conversion rates, precise control over reaction kinetics, and minimal back-mixing. Their design makes them well-suited for gas-phase reactions, such as those used in the production of ammonia, methanol, and hydrogen, where maintaining a controlled concentration gradient is great for optimizing yield. PFRs also play a role in catalytic cracking, a key process in the petroleum industry used to break down large hydrocarbon molecules into more valuable products like gasoline and diesel. The unidirectional flow pattern of PFRs supports high reaction efficiency by ensuring that reactants undergo a predictable transformation, minimizing side reactions and improving selectivity. PFRs can be designed as packed-bed reactors, where solid catalysts or structured packing materials enhance mass and heat transfer, making them ideal for catalytic and heterogeneous reactions. This adaptability allows PFRs to be used in a wide range of chemical production processes, including oxidation reactions, polymer synthesis, and specialty chemical manufacturing. Their ability to achieve high conversion efficiency with precise kinetic control makes them a core of large-scale industrial applications, particularly in the petrochemical, polymer, and specialty chemical sectors.
Derivation of Plug Flow Reactor Equation
The derivation of the Plug Flow Reactor (PFR) equation is important to understand how PFRs efficiently control chemical reactions by precisely managing reaction kinetics, temperature, and pressure. This breakdown explains the basic principles and mathematical steps involved in formulating the PFR equation, providing insight into how these reactors achieve high conversion rates and selectivity.
A PFR operates on the principle of continuous and uniform flow. Reactants enter at one end and move through the reactor in a plug-like manner, advancing steadily toward the outlet without back-mixing or radial dispersion. This flow pattern is similar to a piston moving through a cylinder, where each segment of fluid retains its chemical identity and progresses at the same velocity. This uniform movement ensures that every segment of reactant experiences the same residence time, maintaining consistent reaction conditions. This consistency is good for precise kinetic control, making PFRs effective for complex reactions that require accurate timing and concentration management.
The operation of a PFR relies on applying mass and heat balance within a differential volume of the reactor. By analyzing a small slice of fluid as it moves through the reactor, it is possible to model the chemical kinetics and predict product distribution accurately. In an ideal PFR, the absence of back-mixing and radial dispersion keeps each fluid segment distinct, preserving its chemical history. This unidirectional flow pattern creates a concentration gradient along the reactor’s length, reactant concentrations decrease while product concentrations increase. This gradient efficiently drives the chemical reaction forward. Mass balance is used to track the consumption of reactants and the formation of products, while heat balance accounts for temperature changes that affect reaction rates. For simplicity, the derivation often assumes isothermal conditions, focusing on mass balance and reaction kinetics without the complexity of temperature variations.
The PFR equation is derived from the mass balance on a differential volume of the reactor. Under isothermal conditions, the change in reactant concentration along the reactor’s length is balanced by the reaction rate. The differential form of the mass balance equation is:
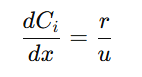
Where:
𝐶𝑖 = Concentration of reactant 𝑖 at position 𝑥
𝑥 = Position along the reactor’s length
𝑟 = Reaction rate, which depends on reactant concentrations and kinetics
𝑢 = Constant fluid velocity
This equation shows that the change in reactant concentration is directly related to the rate of reaction and the velocity of the fluid moving through the reactor. It provides a precise representation of the reaction kinetics within the PFR. To solve this differential equation, boundary conditions are set at the reactor’s inlet, where the initial concentration of each reactant is known:
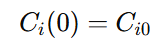
The integration is carried out from the inlet (𝑥=0) to the outlet (𝑥=𝐿), where 𝐿 is the total length of the reactor. Solving the equation across this length gives the concentration profile of each reactant as it moves through the reactor. This concentration profile allows the prediction of exit concentrations and overall reactor performance, helping design reactors that achieve the desired product yield and selectivity.
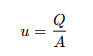
By maintaining a constant velocity, the reactor ensures that all fluid segments have the same residence time for achieving consistent reaction conditions. The mean residence time (𝜏), an important design and operation parameter, is calculated by:
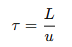
This value represents the average time a reactant spends in the reactor. It is good for tuning the reactor’s performance, especially for fast reactions or those sensitive to exposure time. By adjusting 𝜏, you can optimize conversion rates and product yields.
First-Order Irreversible Reaction in a Plug Flow Reactor
A first-order irreversible reaction in a Plug Flow Reactor (PFR) involves a single reactant decomposing into a product without any backward reaction. This reaction type is important in chemical engineering, as it models many decomposition and isomerization processes. In this example, reactant 𝐴 is converted into product 𝐵 following first-order kinetics. This section explains how the PFR’s design enhances the reaction’s efficiency and selectivity.
In a PFR, the reaction is represented as:
𝐴 → 𝐵
This indicates that reactant 𝐴 irreversibly transforms into product 𝐵 according to first-order kinetics, meaning the reaction rate is directly proportional to the concentration of 𝐴. This simple model is widely used in industrial chemical processes to ensure complete conversion of 𝐴 to 𝐵 under the right conditions. The PFR’s continuous, unidirectional flow with no back-mixing provides an ideal environment for first-order reactions. Each plug of reactant moves through the reactor at a constant velocity, maintaining its chemical identity and experiencing the same reaction conditions. This consistency supports high conversion efficiency and precise control over product selectivity. The governing equation for a first-order irreversible reaction in a PFR is derived from a mass balance on a differential reactor segment. It is expressed as:
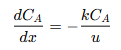
Where:
𝐶𝐴 = Concentration of reactant 𝐴 at position 𝑥
𝑥 = Position along the reactor’s length
𝑘 = Reaction rate constant, indicating the speed of the reaction
𝑢 = Constant fluid velocity through the reactor
This equation shows that the rate of change of 𝐴’s concentration along the reactor’s length depends on the first-order kinetics, where the negative sign indicates the consumption of 𝐴. The absence of back-mixing in the PFR ensures that each differential segment behaves like a small batch reactor, simplifying the mathematical modeling of the reaction. To find the concentration profile of 𝐴 along the reactor, the differential equation is integrated with the boundary condition that the initial concentration at the inlet is known:
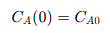
The solution to the differential equation is:
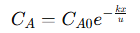
This equation describes how the concentration of 𝐴 decreases exponentially as it moves through the reactor. The rate of this exponential decay depends on the reaction rate constant (𝑘) and the fluid velocity (𝑢). This relationship shows how reactor design parameters such as length (𝐿), flow rate, and reaction rate constant, affect the concentration profile. For example, increasing the reactor length or decreasing the flow rate provides more residence time, allowing more 𝐴 to convert into 𝐵. Conversely, a higher flow rate reduces the contact time, resulting in lower conversion. The overall conversion efficiency (𝑋) of reactant 𝐴 is defined as the fraction of 𝐴 that reacts along the reactor’s length:
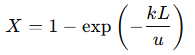
Where:
𝐿 = Total length of the reactor
𝑋 = Conversion efficiency of 𝐴
This equation shows that increasing the reactor length (𝐿) or the rate constant (𝑘) improves conversion efficiency, while a higher fluid velocity (𝑢) reduces it. This formula also illustrates the benefit of the plug flow pattern: the uniform residence time ensures that all reactant plugs experience the same reaction conditions, leading to predictable and consistent conversion rates.
Second-Order Irreversible Reaction in a Plug Flow Reactor
A second-order irreversible reaction in a Plug Flow Reactor (PFR) involves a reaction rate that depends on the square of the reactant’s concentration. This type of reaction is common in processes like polymerization and dimerization. Understanding how PFRs handle these reactions helps design efficient reactors with high conversion rates and selectivity. In a PFR, a typical second-order irreversible reaction involves two molecules of reactant 𝐴 combining to form a product 𝐵, represented as:
2A → B
This reaction is irreversible, meaning once 𝐴 is converted into 𝐵, it does not revert. The PFR’s continuous, uniform flow pattern enhances this conversion by maintaining consistent velocity and residence time throughout the reactor. This design ensures that each plug of reactant experiences the same reaction conditions, maximizing conversion efficiency. The governing equation for this reaction is derived from a mass balance on a small segment of the reactor, assuming no back-mixing or radial dispersion:
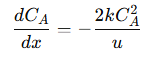
Where:
𝐶𝐴 = Concentration of reactant 𝐴 at position 𝑥
𝑥 = Position along the reactor’s length
𝑘 = Rate constant, indicating the reaction speed
𝑢 = Constant fluid velocity
The factor of 2 accounts for the stoichiometry, where two moles of 𝐴 are consumed to form one mole of 𝐵.
This equation shows that the rate of change of 𝐴’s concentration is proportional to the square of its current concentration, a hallmark of second-order kinetics. This leads to a rapid decrease in 𝐴 as the reaction progresses, especially at high concentrations. To find the concentration profile of 𝐴 along the reactor, the differential equation is integrated from the inlet, where the initial concentration is 𝐶𝐴0, to any point 𝑥 along the reactor:
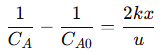
This solution shows that 𝐴’s concentration decreases hyperbolically, unlike the exponential decay seen in first-order reactions. The rate of this decline depends on the reaction rate constant (𝑘), the reactor length (𝑥), and the fluid velocity (𝑢). This relationship emphasizes the impact of operational parameters on reactant concentration and overall conversion efficiency. For example:
• Increasing the reactor length (𝑥) or the rate constant (𝑘) enhances conversion by providing more time or accelerating the reaction.
• Higher fluid velocity (𝑢) reduces residence time, leading to lower conversion.
This model helps predict how adjustments in reactor design and operating conditions influence product yield and selectivity.
The overall conversion efficiency (𝑋) of reactant 𝐴 is calculated as the fraction of 𝐴 transformed into product 𝐵 along the reactor’s length:
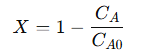
Substituting the integrated concentration profile gives:
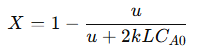
Where:
𝐿 = Total length of the reactor
𝐶𝐴0 = Initial concentration of 𝐴
𝑋 = Conversion efficiency of 𝐴
This equation shows that conversion efficiency increases with:
• Longer reactor length (𝐿), which increases residence time.
• Higher initial concentration (𝐶𝐴0), which boosts the reaction rate.
• Larger rate constant (𝑘), which speeds up the reaction.
Conversely, higher fluid velocity (𝑢) decreases conversion by reducing the time reactants spend in the reactor. These insights allow to optimize reactor performance by adjusting flow rates, reactor length, and operating temperatures (which affect 𝑘).
Understanding of second-order irreversible reaction behavior is good for designing Plug Flow Reactors (PFRs) to maximize product yield while minimizing reactant waste. Since reaction rates in second-order kinetics depend on the concentration of two reactants, optimizing reactor parameters can impact overall conversion efficiency. One key factor influencing conversion is reactor length by extending the length of the PFR, reactants have a longer residence time, allowing the reaction to proceed further toward completion before exiting the system. Similarly, decreasing the flow rate increases residence time, providing more opportunity for reactants to interact and form desired products. Another parameter is temperature, which directly affects the reaction rate constant (𝑘). Increasing the temperature raises the rate constant, thereby accelerating the reaction and improving conversion efficiency. However, you must carefully balance temperature adjustments to avoid unwanted side reactions or thermal degradation of sensitive compounds. These principles allow for precise control over reaction conditions, enabling industries to fine-tune PFR operations for specific production goals.
The ability to control second-order reaction kinetics with precision is advantageous in industries where product quality, consistency, and efficiency are great. In polymerization processes, for example, maintaining optimal monomer concentrations throughout the reactor ensures that polymers achieve consistent molecular weight distributions and desirable physical properties. Even slight variations in concentration or reaction conditions can lead to inconsistencies in polymer structure, affecting material strength, elasticity, or durability. Likewise, in fine chemical and pharmaceutical manufacturing, reaction selectivity and purity are great to ensuring product effectiveness and safety.
Mass Balance in Plug Flow Reactor at Steady State
Analyzing mass balance in a Plug Flow Reactor (PFR) under steady-state conditions is good for understanding its efficiency and optimizing performance across various chemical processes. In a PFR, reactants flow continuously and uniformly from the inlet to the outlet without back-mixing or radial dispersion. This consistent flow pattern allows precise control over reactant concentrations and reaction rates, making PFRs highly effective for complex chemical reactions.
Continuous and Uniform Flow Dynamics
In a plug flow reactor (PFR), the movement of reactants follows a steady, unidirectional path, ensuring that each individual fluid element retains its chemical identity and reaction history as it progresses through the reactor. Unlike reactors that promote mixing, such as continuous stirred-tank reactors (CSTRs), a PFR operates under the principle that every segment of fluid remains distinct, preventing lateral mixing with adjacent segments. This characteristic means that reactant concentrations and reaction extents vary continuously along the reactor's length, rather than being averaged out through turbulent mixing. Because of this plug-like movement, reactants undergo reaction under well-defined conditions at every point in the reactor, allowing for a more controlled and predictable transformation from reactants to products. The resulting concentration and temperature gradients along the length of the reactor play a role in governing the reaction kinetics and overall efficiency of the process.
The uniform and predictable nature of flow in a PFR offers distinct advantages, particularly for reactions that are highly sensitive to fluctuations in concentration or temperature. By eliminating back-mixing, the reactor ensures that the residence time for each reactant molecule is precisely controlled, allowing for more accurate predictions of reaction progress and product formation. This feature is useful in optimizing product selectivity, as certain reactions may favor specific conditions that can be carefully maintained throughout the reactor's length. The absence of internal mixing simplifies mathematical modeling, making it easier to develop kinetic models and design reactors for maximum efficiency. This streamlined approach to reaction engineering not only enhances performance but also reduces the complexity associated with scaling up processes for industrial applications.
Deriving the Mass Balance Equation
The mass balance equation for a PFR is derived by analyzing a small differential volume (𝑑𝑉) of the reactor. This approach considers the flow of reactant 𝐴 into and out of this volume, along with the rate at which it reacts. Under steady-state conditions, there is no accumulation of 𝐴 within 𝑑𝑉, meaning the inflow rate equals the outflow rate adjusted for the amount consumed by the reaction. The general mass balance equation is:
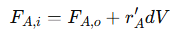
Where:
𝐹𝐴,𝑖 = Molar flow rate of reactant 𝐴 entering the differential volume
𝐹𝐴,𝑜 = Molar flow rate of 𝐴 leaving the differential volume
𝑟′𝐴 = Reaction rate per unit volume for 𝐴
𝑑𝑉 = Differential volume element
This equation shows that the decrease in the flow rate of 𝐴 is due to its consumption within the volume 𝑑𝑉. Since there is no back-mixing, each plug of reactant retains its concentration as it moves forward, maintaining the plug flow pattern.
Application of the Continuity Equation
To relate the outlet molar flow rate to the inlet flow rate, the continuity equation is used:
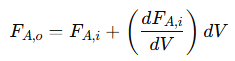
Substituting this into the mass balance equation gives:
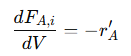
This shows that the change in molar flow rate of 𝐴 is directly proportional to the reaction rate. The negative sign indicates the consumption of 𝐴 as it is converted into products. This relationship highlights how the reaction rate affects the concentration profile of 𝐴 along the reactor’s length. Integrating this differential equation along the length of the reactor provides the general expression for the reaction rate of 𝐴:
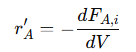
This shows that the reaction rate is proportional to the change in molar flow rate of 𝐴 along the reactor. The negative sign reflects the decrease in 𝐴 as it reacts to form products. This integrated form of the mass balance equation allows to calculate the concentration profile of 𝐴 throughout the reactor, enabling accurate predictions of product yields and overall conversion efficiency.
Extension to Complex Reactions
For more complex reactions, such as a bimolecular reaction 𝐴+𝐵→𝐶+𝐷, the mass balance equations can be extended to account for all reactants and products. For each species, the differential equations are:
• For reactant 𝐴:
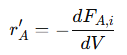
• For reactant 𝐵:
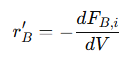
• For product 𝐶:
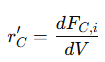
• For product 𝐷:
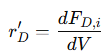
Negative signs indicate consumption of reactants, while positive signs denote formation of products. These equations provide a complete mass balance for complex reaction networks, allowing precise modeling of reaction kinetics and product distribution.
Plug Flow Reactor with Constant Volumetric Flow
In a Plug Flow Reactor (PFR), assuming no volumetric flow variation simplifies the mathematical modeling of chemical reactions. This assumption is useful for gas-phase reactions under constant temperature and pressure (isothermal and isobaric conditions) and for liquid-phase reactions where the fluid density remains constant. By maintaining a steady flow rate throughout the reactor, calculations for reaction kinetics, conversion efficiency, and product distribution become more straightforward and reliable.
Assuming that the volumetric flow rate (𝑄) remains constant throughout the PFR simplifies the analysis of chemical reactions. This constant flow ensures that the fluid velocity is uniform along the entire reactor length, leading to consistent residence times for all fluid elements. This uniformity is good for maintaining the plug flow pattern, where each segment of fluid moves forward like a piston, without back-mixing or radial dispersion.
This steady flow pattern ensures that changes in reactant concentrations occur only in the axial direction, efficiently driving the chemical reaction forward. It also preserves the chemical identity of each fluid segment, allowing precise control over reaction conditions and kinetics. This is advantageous for reactions requiring accurate residence time and temperature control. Under the assumption of constant volumetric flow, the flow rate at any point in the reactor remains the same from the inlet to the outlet:
Q i = Qo =Q
This steady-state condition means that the flow parameters are consistent throughout the reactor’s length, simplifying the mass balance equation. It allows the analysis to focus on molar flow rates and concentration changes of reactants and products without considering variations in velocity or volume. The molar flow rate of a reactant 𝐴 at any position 𝑥 along the reactor is given by:
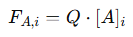
Where:
𝐹𝐴,𝑖 = Molar flow rate of 𝐴 entering the differential volume element 𝑑𝑉
[A]i = Concentration of 𝐴 at position 𝑥
𝑄 = Constant volumetric flow rate
This equation links the molar flow rate of reactant 𝐴 to its concentration and the volumetric flow rate. Because 𝑄 is constant, changes in 𝐹𝐴,𝑖 are directly related to changes in [𝐴]𝑖. The reaction rate for reactant 𝐴, denoted by 𝑟′𝐴, describes the rate at which 𝐴 is consumed along the reactor’s length. It is calculated using the differential change in the molar flow rate:
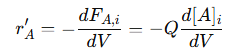
The negative sign indicates the consumption of reactant 𝐴 as it is transformed into products. This relationship shows that the reaction rate is directly proportional to the concentration gradient along the reactor. Because 𝑄 is constant, the concentration changes only in the axial direction, ensuring that the reaction proceeds smoothly from the inlet to the outlet. This unidirectional flow pattern helps maintain consistent reaction conditions, leading to predictable product yields and high conversion efficiency. By integrating this differential equation along the length of the reactor, it is possible to calculate how the concentration of 𝐴 changes from the inlet to the outlet:
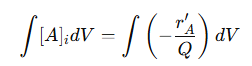
This integration provides a concentration profile of 𝐴 along the reactor, revealing how much reactant is consumed and how much product is formed. It also shows the impact of reactor design parameters such as length, flow rate, and reaction rate constant on conversion efficiency and product distribution. Keeping mathematical models simple means fewer variables, making calculations easier and simulations faster. Accurate concentration predictions help design reactors for maximum efficiency and selectivity, ensuring optimal performance. Consistent residence times and flow rates allow for precise process control, leading to reliable product quality. Overall, assuming no volumetric flow variation in PFRs simplifies chemical reaction modeling while maintaining accuracy, making it an important tool for efficient reactor design and operation.
Advantages and Disadvantages of Plug Flow Reactors
Plug Flow Reactors (PFRs) are widely used in chemical processing due to their unique design, which ensures the continuous and uniform movement of reactants. This design allows precise control over reaction kinetics and residence times, making PFRs highly efficient for many chemical processes. However, they also have limitations that must be considered depending on the application. This section examines the advantages and disadvantages of PFRs.
Advantages of Plug Flow Reactors
High Conversion Efficiency
Plug Flow Reactors (PFRs) are known for their high conversion efficiency, which results from their unique flow characteristics. Unlike other reactor types, PFRs ensure that reactants move in a single direction without any back-mixing. This means that once a fluid element enters the reactor, it continues to flow forward without blending with other portions of the fluid. Because of this controlled movement, the chemical composition at any given point in the reactor remains distinct and predictable. One major advantage of this steady flow is that it minimizes the residence time distribution, meaning all reactant particles spend an equal amount of time inside the reactor. As a result, the reaction occurs uniformly throughout the entire length of the reactor, improving efficiency and making PFRs useful for processes that require strict control over reaction conditions.
Another reason for the high conversion efficiency of PFRs is their ability to maintain a stable concentration gradient throughout the reactor. Since reactants are continuously consumed as they progress through the reactor, the concentration gradually decreases along the flow path, which can help drive reactions to completion more effectively. This makes PFRs more efficient than Continuous Stirred Tank Reactors (CSTRs), where constant mixing can lead to variations in concentration that may reduce reaction efficiency. For reactions that are highly sensitive to concentration changes or require precise timing, PFRs offer an advantage. By ensuring a uniform reaction environment, they help optimize reaction rates, minimize unwanted byproducts, and maximize product yield.
Enhanced Reaction Selectivity
The structured flow in plug flow reactors (PFRs) ensures that reactants are consistently exposed to the same reaction conditions as they move through the reactor. This steady and uniform exposure plays a role in minimizing the formation of unwanted by-products, which can negatively impact the efficiency of the reaction. By reducing these undesirable side reactions, PFRs help improve the overall selectivity of the reaction, making them useful for chemical processes that require high precision. This is important in industries where even small variations in chemical composition can lead to differences in product performance, such as in the manufacturing of specialty chemicals and pharmaceutical products. Because each plug of reactant moves at the same velocity and experiences identical reaction conditions, the reaction proceeds in a controlled and predictable manner. This level of consistency ensures that the desired chemical transformation occurs as intended, resulting in a higher yield of the target product while minimizing the need for additional purification steps.
The ability of PFRs to provide a uniform reaction environment directly contributes to enhanced product consistency and improved reaction efficiency. Since every portion of the reactant stream follows the same pathway and undergoes the same reaction conditions, the final product exhibits reliable and repeatable characteristics. This is beneficial for processes where maintaining strict quality control is important, as even slight variations in temperature, pressure, or reactant concentration could alter the final outcome. The high degree of selectivity in PFRs makes them ideal for reactions that involve sensitive intermediate species, where precise control over reaction kinetics is necessary. Additionally, because PFRs operate with a continuous flow, they can be easily scaled up for industrial applications without compromising reaction efficiency. This scalability, combined with their ability to enhance reaction selectivity, makes PFRs a preferred choice for many chemical production processes, ensuring a consistent, high-quality output in a variety of applications.
Operational and Energy Efficiency
Plug Flow Reactors (PFRs) operate in a continuous manner, which means they do not need to be stopped and restarted frequently. This continuous operation reduces downtime, allowing for higher production rates and improved efficiency in chemical processing. Since PFRs do not require mechanical agitation, they consume less energy compared to other reactor types that rely on moving parts to mix reactants. Lower energy consumption translates to reduced operational costs, making PFRs a cost-effective choice for large-scale production. Their ability to maintain steady-state operation ensures that chemical reactions proceed in a predictable manner, contributing to process stability. The absence of moving components also reduces maintenance needs, minimizing unexpected breakdowns and enhancing overall reliability. Because of these advantages, PFRs are widely used in industries that require consistent, high-throughput chemical processing while keeping energy use and operational costs as low as possible.
Another major advantage of PFRs is their ease of scaling up from laboratory experiments to full industrial production. Since the reaction conditions remain uniform along the reactor’s entire length, increasing production capacity can be achieved by simply extending the length of the reactor or using multiple parallel tubes. This scalability ensures that chemical manufacturers can expand their operations without major modifications to the reactor design. The predictable flow pattern within a PFR also makes it easier to control reaction variables, ensuring consistent product quality across different production scales. Because each portion of the reactants moves through the reactor without back-mixing, the reaction process remains stable and uniform. This feature simplifies process optimization and allows for better control over reaction efficiency. As a result, industries that require precise control over chemical reactions, often choose PFRs for their ability to maintain consistency and reliability on both small and large scales.
Superior Heat and Mass Transfer
The plug flow design helps transfer heat and mass efficiently, which keeps reactions stable, especially in processes that release or absorb a lot of heat. Because there is no back-mixing, heat spreads evenly through the reactor, preventing areas that are too hot or too cold. This even heat distribution keeps reaction rates steady and maintains good product quality. If heat were uneven, it could cause problems like slow reactions or unwanted side products. By keeping the temperature stable, the plug flow design also improves safety, reducing the risk of dangerous overheating in reactions that release a lot of heat. Since the reaction stays under control, it runs more smoothly and efficiently, leading to better results. This design also ensures that the reaction follows a steady and predictable path, which improves efficiency and keeps the product pure. As the reactants move through the reactor, they all go through the same conditions without mixing with other parts. This allows for better control over the process and ensures that the reaction happens as expected. Because of this, fewer unwanted byproducts form, and more of the desired product is made. The plug flow design’s ability to transfer heat well, maintain stable temperatures, and keep reactions controlled makes it very useful for industrial processes where quality and efficiency are important.
Versatility in Applications
Plug Flow Reactors (PFRs) are very useful because they can handle many different types of chemical reactions. They work well for fast reactions, irreversible reactions, and gas-phase reactions because the reactants move smoothly through the reactor in a steady flow. PFRs are also great for reactions that use solid catalysts, as the even flow makes sure all reactants get the same exposure to the catalyst. This helps chemical reactions happen more efficiently and gives more consistent results. Because of these advantages, PFRs are widely used in industries like petrochemicals and pharmaceuticals, where precise control over reactions is needed to make high-quality products. Their ability to keep reactions stable and efficient makes them an important tool for many chemical processes. PFRs are also very helpful in making polymers because they keep monomer concentrations steady throughout the reaction. This is important for producing polymers with uniform properties, such as consistent strength and durability. The smooth flow in a PFR reduces variations in concentration and reaction time, leading to a more reliable final product. Since PFRs operate continuously, they can also save time and improve efficiency compared to batch reactors. Their ability to handle different reactions with accuracy and reliability makes them valuable in many industries. Whether used for catalysis, gas-phase reactions, or polymer production, PFRs offer a dependable and efficient way to control chemical reactions and maintain product quality.
Disadvantages of Plug Flow Reactors
Limited to Specific Reaction Types
Plug Flow Reactors (PFRs) work best for chemical reactions that move in one direction and do not need to reverse or reach balance. They are most effective for irreversible reactions, where reactants turn into products without going back. Since PFRs have a steady flow, the reactants move forward without mixing back with the products. This setup is not ideal for reactions that need to reach equilibrium because the lack of mixing prevents the reaction from adjusting along the way. In these cases, a PFR may not allow the reaction to reach its full potential, making it less effective for certain chemical processes. Some reactions require more time and mixing to get the best results, but PFRs do not provide this flexibility. Because of this, some reactions may not fully complete, leading to lower efficiency. For reactions that need better mixing and longer reaction times, a Continuous Stirred-Tank Reactor (CSTR) or another type of reactor might work better. CSTRs constantly stir the contents, ensuring even mixing and allowing the reaction to move in both directions if needed. This makes them useful for reactions that require careful control to get the best yield. While PFRs are great for certain reactions, they may not be the best choice when a reaction needs more time to balance or requires constant mixing. Choosing the right reactor depends on the type of reaction and the desired outcome.
Sensitivity to Feed Quality and Operating Conditions
Plug Flow Reactors (PFRs) need a steady and well-mixed feed to keep reaction rates and product quality consistent. If the feed changes in concentration, temperature, or pressure, it can affect how well the reactor works. Uneven feed quality may lead to inconsistent conversion rates and variations in the final product. In industries where precise product quality is important, even small changes can cause problems like unwanted side reactions or lower efficiency. To avoid these issues, it is important to keep the feed stable and properly controlled. A well-regulated feed ensures that the reactor operates smoothly and produces reliable results. PFRs are highly sensitive to changes in feed conditions because any variation directly affects how the reaction happens inside the reactor. Since reactions take place as the feed moves through, any change in its properties can lead to uneven processing and unpredictable results. To prevent this, operators must carefully monitor and control the feed. Temperature shifts, pressure changes, or impurities can disturb the reaction and reduce efficiency. Using advanced monitoring systems helps maintain stable conditions, ensuring better performance, higher product quality, and fewer production problems.
Potential for Channeling
In packed-bed plug flow reactors (PFRs), solid catalysts or packing materials help drive chemical reactions. However, channeling can occur when the flow of reactants does not spread evenly through the packed bed. Instead of flowing uniformly, the reactants may take certain paths, skipping parts of the catalyst. This causes an uneven reaction, where some areas work efficiently while others do not. As a result, some reactants pass through the reactor without fully reacting, reducing overall efficiency. Since PFRs need even flow for the best performance, channeling can be a big problem, especially in industries that require high conversion rates. Channeling leads to uneven reaction rates and lower overall efficiency. When parts of the catalyst bed are bypassed, fewer reactants get converted into products, leading to wasted materials and lower yields. To prevent this, you must design reactors carefully. They can use uniform packing, flow distributors, or special structures to spread the flow evenly. Proper design helps minimize channeling, making the reactor more efficient and improving chemical production.
Complexity in Design and Scale-Up
Even though plug flow reactors (PFRs) have a simple design, making them bigger or designing them for large-scale use can be quite tricky. When scale up a PFR, they need to make sure that the flow of materials stays smooth and that the reaction conditions remain the same throughout the reactor. This requires careful calculations to understand how fluids move, how heat spreads, and how fast reactions happen inside the system. These factors become even more important in large industrial settings, where small changes in flow or temperature can affect efficiency and product quality. If a PFR is not scaled up properly, it might not work as well as expected, leading to problems in production. To scale up a PFR, often make it longer or set up multiple reactors side by side. However, simply increasing the size is not enough. They must carefully adjust flow rates to ensure that materials move at the right speed for the reaction to complete. Pressure changes inside the reactor also need to be controlled to prevent flow problems. Another issue is heat transfer since larger reactors may struggle to keep an even temperature, which can cause parts of the reactor to overheat or cool down too much. Many use advanced methods to manage these factors so that the larger reactor works just as well as the original smaller version.
Maintenance and Operational Challenges
Plug flow reactors (PFRs) can develop fouling, which happens when solid materials build up inside the reactor. These deposits can come from leftover reactants, byproducts, or impurities in the feed. Over time, fouling can block the flow of materials and make heat transfer less effective. Since temperature control is important for reactions, this can slow down or change the reaction process, reducing efficiency. To prevent this, regular cleaning is needed, but this can be costly and may require stopping or slowing down operations. If fouling is not managed, it can also cause long-term damage to reactor parts, leading to even more expensive repairs. Besides fouling, PFRs can also face blockages or disruptions in flow. These reactors need a steady movement of materials, so any blockage can cause problems. Blockages may come from solid particles, equipment issues, or changes in the feed composition. When this happens, pressure can change, and the reaction may not complete properly, leading to lower product quality. Uneven flow can also affect temperature and concentration levels inside the reactor, making the process less efficient. To avoid these problems, constant monitoring is needed. Many systems use sensors and controls to track performance, but these add to operational costs. Despite these challenges, regular maintenance and careful monitoring help keep PFRs running smoothly and efficiently.
Applications of Plug Flow Reactors
Plug Flow Reactors (PFRs) are widely used in chemical engineering due to their efficiency and versatility. Their design ensures unidirectional flow and consistent residence times, allowing precise control over reaction kinetics and product distribution. This makes them suitable for various chemical reactions, including gas-phase, liquid-phase, heterogeneous, and homogeneous systems. Their scalability and cost-effectiveness are valuable in large-scale industrial production.
Chemical Manufacturing
Plug Flow Reactors (PFRs) are important in making large amounts of chemicals like sulfuric acid, ethylene oxide, and methanol. Their design allows chemicals to flow continuously, which speeds up reactions and increases production. This is useful for industries that need to make high volumes of chemicals quickly. PFRs provide stable reaction conditions, ensuring that the final product is pure and consistent in quality. By keeping reaction rates steady and reducing unwanted by-products, PFRs help maximize production efficiency. This reliability is great for industries that depend on precise chemical processes to produce large quantities efficiently and at a lower cost. PFRs also make chemical manufacturing more efficient by preventing back-mixing and keeping reactions uniform. Their steady flow allows for better control over reaction conditions, reducing the need for extra purification steps. This lowers production costs and decreases waste, making operations more sustainable. Because of their ability to produce chemicals quickly, consistently, and with minimal waste, PFRs are widely used in industries that require high-volume chemical production. Their design makes them a preferred choice for companies looking to improve efficiency and product quality while keeping costs under control.
Petrochemical Processing
In the petrochemical industry, Plug Flow Reactors (PFRs) are commonly used for refining processes such as catalytic cracking and hydrogenation. Their precise control over reaction conditions allows the efficient production of valuable chemicals like ethylene, propylene, and butadiene, which are used in making plastics and synthetic materials. Because PFRs process high volumes of hydrocarbons continuously, they help refineries operate smoothly and maintain product quality. The unidirectional flow in PFRs prevents back-mixing, preserving the purity of reactants and improving overall efficiency. Another advantage of PFRs in petrochemical processing is their ability to handle heat effectively, which is important for high-temperature refining reactions. By maintaining steady temperature and pressure, PFRs help maximize yields while reducing unwanted by-products. Their design also makes them adaptable for both large-scale refining operations and specialized petrochemical production. Because of their efficiency, reliability, and ability to process large amounts of material without interruption, PFRs play a role in the petrochemical industry, helping refineries meet global demand while improving energy efficiency.
Pharmaceutical Production
Plug Flow Reactors (PFRs) are widely used in making medicines, especially in the production of active pharmaceutical ingredients (APIs). Their ability to maintain precise reaction conditions ensures that medicines are produced with high purity and consistency. Because all molecules in a PFR experience the same reaction conditions, there are fewer variations in product quality. This is important in the pharmaceutical industry, where strict regulations require consistent and high-quality drug production. By reducing the formation of unwanted by-products, PFRs also help maximize yield, ensuring that expensive raw materials are used efficiently. PFRs also improve pharmaceutical manufacturing by enabling continuous production instead of batch processing. This reduces variability between different production runs and increases efficiency. The steady flow of reactants in PFRs makes them ideal for producing specialty drugs that require precise control over reaction times and conditions. PFRs support modern manufacturing techniques like continuous flow chemistry, which helps companies reduce waste and lower costs while meeting strict quality and safety standards. Because of these benefits, PFRs are an important technology in pharmaceutical production.
Catalytic Processes
Plug Flow Reactors (PFRs) are highly effective in catalytic reactions, where reactants must have even contact with catalysts for the best results. Their steady flow ensures that reactants pass uniformly over catalytic surfaces, improving reaction speed and efficiency. This is useful in industrial processes like hydrogenation and oxidation, where precise reaction control leads to higher yields. Since PFRs maintain stable conditions, catalysts last longer, reducing costs and improving overall performance. By preventing uneven mixing, PFRs also reduce unwanted side reactions, making them a preferred choice for industries that rely on catalytic processes. PFRs also enhance large-scale catalytic production by optimizing reaction conditions and reducing waste. Their design allows better temperature and pressure control, leading to more efficient use of catalysts. This is important for industries such as petrochemicals, pharmaceuticals, and fine chemicals, where even small improvements in efficiency can impact costs and product quality. Because of their ability to maximize catalyst performance while minimizing waste and energy use, PFRs play a role in industrial catalytic reactions.
Environmental Applications
Plug Flow Reactors (PFRs) are important in environmental protection, especially for reducing pollution and treating industrial waste. They are commonly used in flue gas treatment, where harmful emissions like sulfur dioxide and nitrogen oxides are converted into safer compounds. PFRs also play a role in wastewater treatment by breaking down hazardous chemicals into less harmful by-products. Their continuous flow design ensures efficient pollutant removal, helping industries meet environmental regulations while reducing operational costs. In addition to pollution control, PFRs support sustainability by improving energy efficiency and reducing waste. Their ability to process large volumes of industrial waste continuously makes them a reliable solution for large-scale environmental management. By incorporating PFR technology, industries can reduce their environmental impact while maintaining efficient production. As concerns about pollution and climate change grow, PFRs are becoming an increasingly valuable tool for industries seeking cleaner and more sustainable manufacturing solutions.
Versatility in Heterogeneous and Homogeneous Reactions
PFRs can handle both heterogeneous and homogeneous reactions, making them highly versatile in chemical synthesis. They are useful for complex organic syntheses requiring precise thermal control and material handling.In heterogeneous reactions, the uniform flow pattern ensures even contact between reactants and solid catalysts, maximizing conversion rates and product selectivity. In homogeneous reactions, the consistent residence times maintain steady reaction kinetics, enhancing yield and product quality. This adaptability makes PFRs suitable for a wide range of chemical processes, from basic commodity chemicals to complex fine chemicals and pharmaceuticals. Their precise control over reaction conditions allows the synthesis of high-purity products with minimal by-products.
Integral in Fine Chemical Production
PFRs are use in producing fine chemicals, where high purity and specific reaction pathways are required. They provide precise control over temperature, pressure, and residence time, ensuring consistent product quality and yield. This precision is great in manufacturing dyes, fragrances, specialty coatings, and other fine chemicals that require strict quality standards. PFRs minimize by-product formation and enhance reaction selectivity, optimizing raw material usage and reducing waste. Their scalability also allows fine chemical manufacturers to efficiently transition from small-scale laboratory synthesis to full-scale industrial production while maintaining product consistency.
Conclusion
Plug Flow Reactors are super useful for managing chemical reactions with precision. They work by keeping everything moving in one direction, which helps get more product out of the same amount of chemicals and keeps things safe and consistent. They're especially good for big industrial jobs where you need to make a lot of product without wasting materials or energy. However, they're not perfect for every situation, sometimes they don't mix well enough for certain reactions, and they can be sensitive to changes in the materials they use. But overall, PFRs are a go-to choice for a lot of manufacturing processes because they balance efficiency with effectiveness, keeping industries running smoothly and economically.
About us
ALLELCO LIMITED
Read more
Quick inquiry
Please send an inquiry, we will respond immediately.
Frequently Asked Questions [FAQ]
1. What is the principle of plug flow reactor?
The principle of a plug flow reactor (PFR) is that the reactants flow through a cylindrical pipe in a manner where all elements of the fluid move through the reactor at the same velocity, with no mixing in the flow direction. This results in each segment of the fluid entering and exiting the reactor at different times, essentially maintaining a "plug" of fluid that moves through the reactor. The concentration and reaction rate vary along the length of the reactor but are constant across any cross-section perpendicular to the flow.
2. What is the PFR feature?
A key feature of a plug flow reactor is its ability to maintain a steady gradient of reactant concentration from the inlet to the outlet. This ensures that the reaction progresses as a series of infinitesimally small batch reactors sequenced along the length of the tube. This setup provides precise control over reaction times and conditions, which is ideal for reactions that are sensitive to residence time.
3. What are the industrial applications of PFR?
Plug flow reactors (PFRs) are widely used in industries where precise control over reaction times is good for efficiency and product quality. In chemical production, PFRs allow for the continuous creation of chemicals, ensuring uniform product characteristics through stable flow and reaction conditions. They are important in petrochemical processes, such as hydrocarbon cracking to produce ethylene and other olefins, where exact timing and temperatures are key to maximizing yields and reducing by-products. In pharmaceutical manufacturing, PFRs are important for drug synthesis. The precise control they offer over reaction times and conditions helps ensure that pharmaceuticals are pure and yield sufficiently, which is best for patient safety and drug effectiveness. PFRs are also used in water treatment, specifically in the chlorination of water, to effectively disinfect while controlling chemical exposure and minimizing excess use.
4. How does a plug flow digester work?
A plug flow digester operates on the principle similar to a PFR but is specifically designed for the anaerobic digestion of organic materials. In this setup, organic waste is continuously fed into one end of a sealed, horizontal reactor. As the waste moves through the reactor, microorganisms digest the organic matter, producing biogas (methane and carbon dioxide). The digester maintains a uniform flow that prevents mixing and ensures that each segment of waste has a consistent retention time, optimizing gas production and minimizing undigested waste.
5. What are the benefits of PFR?
Using a plug flow reactor (PFR) improves chemical processes by offering high conversion efficiency, meaning it uses all reactants effectively to maximize output. PFRs maintain precise temperature control, boosting reaction rates and yields. They are scalable, allowing easy transition from small-scale lab setups to large-scale industrial production without affecting the process quality. PFRs prevent back-mixing, need for processes that require sequential reactions without interference. They speed up production by keeping reactants moving continuously towards completion, reducing overall reaction time and cutting costs.

What Are Infrared Sensors and How Do They Work
on March 4th

AD5340BRUZ Power Consumption and Performance Review
on March 3th
Popular Posts
-

Complex Instruction Set Computers: How They Changed Computing?
on April 18th 147749
-

USB-C Pinout and Features
on April 18th 111904
-
Using Xilinx Unified Simulation Primitives: A Comprehensive Guide to FPGA Design and Simulation
on April 18th 111349
-

Power Supply Voltages in Electronics: Meaning of VCC, VDD, VEE, VSS, and GND
on April 18th 83714
-

RJ45 Connector Guide: Pinout, Wiring, Cable Types, and Uses
on January 1th 79502
-

The Ultimate Guide to Wire Color Codes in Modern Electrical Systems
The way our electrical systems use colors isn’t just for looks. Each wire color now indicates a specific function, making it easier to identify and handle electrical components correctly during ins...on January 1th 66868
-

Quality (Q) Factor: Equations and Applications
The quality factor, or 'Q', is important when checking how well inductors and resonators work in electronic systems that use radio frequencies (RF). 'Q' measures how well a circuit minimizes energy...on January 1th 63004
-

Purge Valve Guide: Function, Symptoms, Testing, and Replacement for Optimal Engine Performance
The purge valve is a key part of a car’s system that helps keep the air clean by managing fuel vapors before they can escape into the atmosphere. This not only helps the environment by reducing pol...on January 1th 62938
-

Achieving Peak Performance with the Maximum Power Transfer Theorem
The Maximum Power Transfer Theorem explains how energy from a source, such as a battery or generator, flows to a connected load. It shows the exact condition where the load receives the most power....on January 1th 54076
-

A23 Battery Specifications and Compatibility
The A23 battery is a small, cylinder-shaped battery with high voltage. Also called 23A, 23AE, or MN21, it runs at 12 volts and much higher than AA or AAA batteries. Its special design make...on January 1th 52087















































